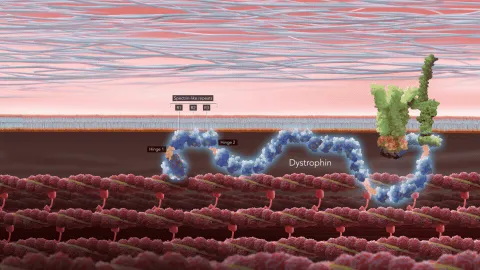
Important Safety Information, including Boxed Warning
BOXED WARNING: Acute Serious Liver Injury and Acute Liver Failure
Acute serious liver injury, including life-threatening and fatal acute liver failure, has occurred.
Patients with preexisting liver impairment may be at higher risk.
Prior to infusion, assess liver function by clinical examination and laboratory testing. Administer systemic corticosteroids before and after ELEVIDYS infusion. Continue to monitor liver function weekly for the first 3 months after infusion and continue until results are unremarkable.
Instruct patients to maintain proximity to an appropriate healthcare facility, as determined by the healthcare provider, for at least 2 months following ELEVIDYS infusion.
Obtain prompt consultation with a specialist (e.g., gastroenterologist or hepatologist) if acute serious liver injury or impending acute liver failure is suspected.
CONTRAINDICATION
ELEVIDYS is contraindicated in patients with any deletion in exon 8 and/or exon 9, including a deletion of any portion or the entirety of these exons, in the DMD gene.
WARNINGS AND PRECAUTIONS
Acute Serious Liver Injury and Acute Liver Failure
See Boxed Warning.
- Acute serious liver injury marked by elevations of liver enzymes (e.g., GGT, ALT) and total bilirubin and acute liver failure has occurred with ELEVIDYS. Onset of the liver injury typically begins within 8 weeks of ELEVIDYS administration. In non-ambulatory patients treated with ELEVIDYS, acute liver failure with fatal outcome has occurred in the clinical and post-marketing settings.
- Life-threatening mesenteric vein thrombosis, complicated by bowel ischemia and necrosis, and portal hypertension have been reported following acute liver injury associated with ELEVIDYS in a non-ambulatory patient.
- Patients with preexisting liver impairment, chronic hepatic condition, or acute liver disease (e.g., acute hepatic viral infection) may be at higher risk of acute serious liver injury or acute liver failure. Postpone ELEVIDYS administration in patients with acute liver disease until resolved or controlled.
- Systemic corticosteroid treatment is recommended for patients before and after ELEVIDYS infusion. Adjust corticosteroid regimen when indicated.
Serious infections
- Increased susceptibility to serious infections may occur due to concomitant administration of corticosteroid regimen and additional immunosuppressants, and ELEVIDYS. Serious respiratory infections, including with fatal outcomes, have occurred in patients taking immunosuppressant corticosteroids required for ELEVIDYS administration.
- Monitor patients for signs and symptoms of infection before and after ELEVIDYS administration and treat appropriately.
- Administer immunizations according to best clinical practices and immunization guidelines prior to initiation of the corticosteroid regimen required before ELEVIDYS infusion.
- Avoid administration of ELEVIDYS to patients with active infections.
Myocarditis
- Acute, serious, life-threatening myocarditis and troponin-I elevations have been observed within 24 hours to more than 1 year following ELEVIDYS infusion.
- If a patient experiences myocarditis, those with pre-existing left ventricle ejection fraction (LVEF) impairment may be at higher risk of adverse outcomes.
- Monitor troponin-I before ELEVIDYS infusion and weekly for the first month following infusion and continue monitoring if clinically indicated, until results return to near baseline levels or stabilize.
- More frequent monitoring may be warranted in the presence of cardiac symptoms, such as chest pain or shortness of breath.
- Advise patients to contact a physician immediately if they experience cardiac symptoms.
Infusion-related Reactions
- Infusion-related reactions, including hypersensitivity reactions and anaphylaxis, have occurred during or up to several hours following ELEVIDYS administration. Closely monitor patients during and for at least 3 hours after the end of infusion. If symptoms of infusion-related reactions occur, slow or stop the infusion and give appropriate treatment. Once symptoms resolve, the infusion may be restarted at a lower rate.
- ELEVIDYS should be administered in a setting where treatment for infusion-related reactions is immediately available.
- Discontinue infusion for anaphylaxis.
Immune-mediated Myositis
- Immune-mediated myositis, including serious and life-threatening events, has occurred approximately 1 month following ELEVIDYS infusion. Signs and symptoms include severe muscle weakness, including dysphagia, dyspnea, dysphonia, and hypophonia.
- Severe to life-threatening immune-mediated myositis has been reported in patients with deletions including portions of exons 1-17 and/or exons 59-71 of the DMD gene.
- Regardless of genetic mutation, advise patients to contact a physician immediately if they experience any unexplained increased muscle pain, tenderness, or weakness, including dysphagia, dyspnea, dysphonia, or hypophonia, as these may be symptoms of myositis. Consider additional immunomodulatory treatment based on patient's clinical presentation and medical history if these symptoms occur.
Preexisting Immunity against AAVrh74
- In AAV-vector based gene therapies, preexisting anti-AAV antibodies may impede transgene expression at desired therapeutic levels. Following treatment with ELEVIDYS, all patients developed anti-AAVrh74 antibodies.
- Perform baseline testing for the presence of anti-AAVrh74 total binding antibodies prior to ELEVIDYS administration.
- ELEVIDYS administration is not recommended in patients with elevated anti-AAVrh74 total binding antibody titers ≥1:400.
ADVERSE REACTIONS
- The most common adverse reactions (incidence ≥5%) reported in clinical studies were vomiting, nausea, liver injury, pyrexia, thrombocytopenia, and troponin-I increased.
Report negative side effects of prescription drugs to the FDA. Visit www.fda.gov/medwatch or call 1-800-FDA-1088. You may also report side effects to Sarepta Therapeutics at 1-888-SAREPTA (1-888-727-3782).
Please see full Prescribing Information, including Boxed Warning, and Medication Guide.